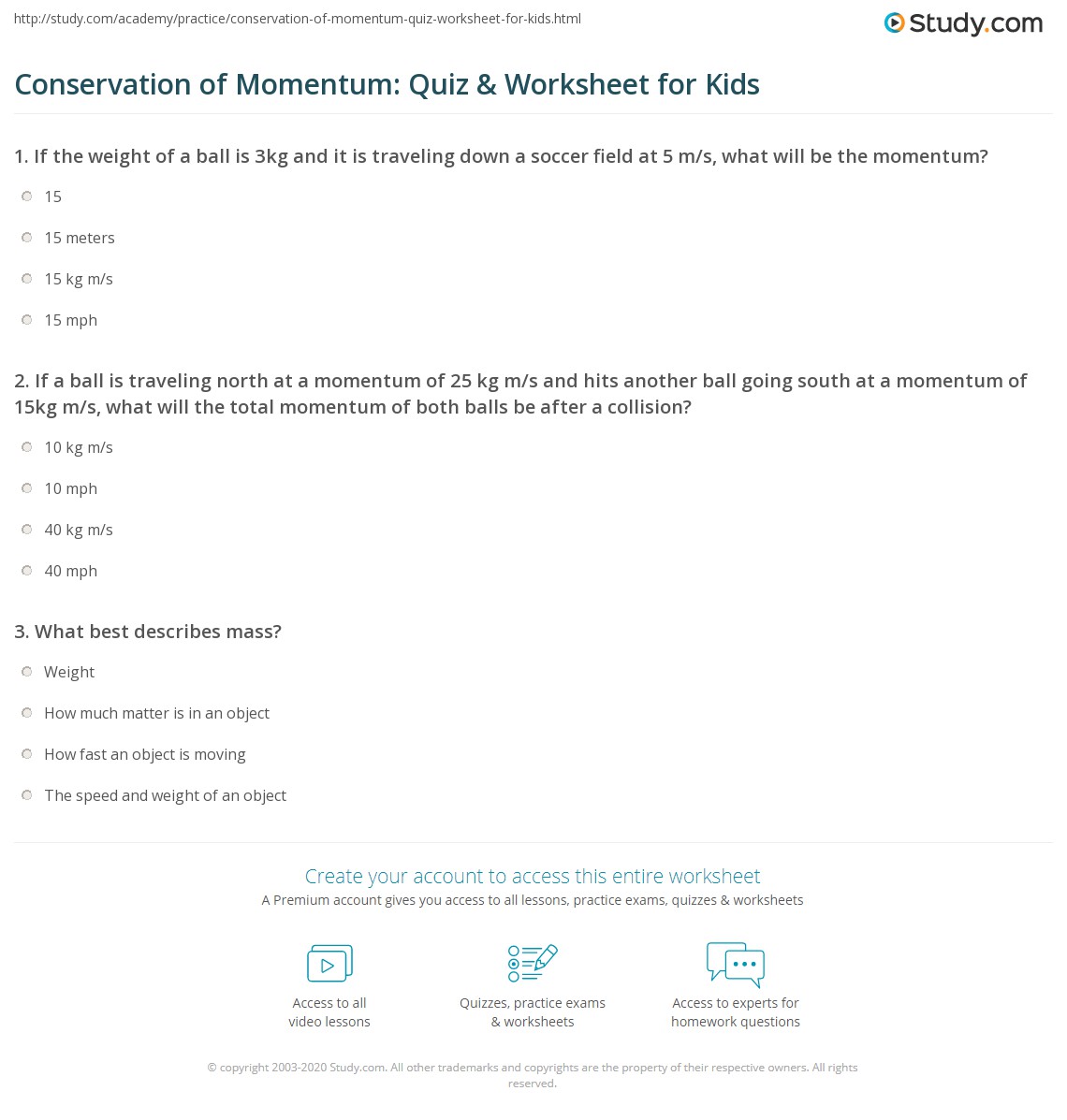
Law of conservation of mass lab pdf
Law of conservation of mass lab pdf
In this lab, students will conduct a chemical reaction that will be used to launch a rocket. Grade Level Elementary school Objectives By the end of this lab, students should understand that mixing chemicals causes a reaction. be aware of the law of conservation of matter. be able to describe gas pressure and connect it to their observations in the experiment. be able to discuss Newton‟s Law
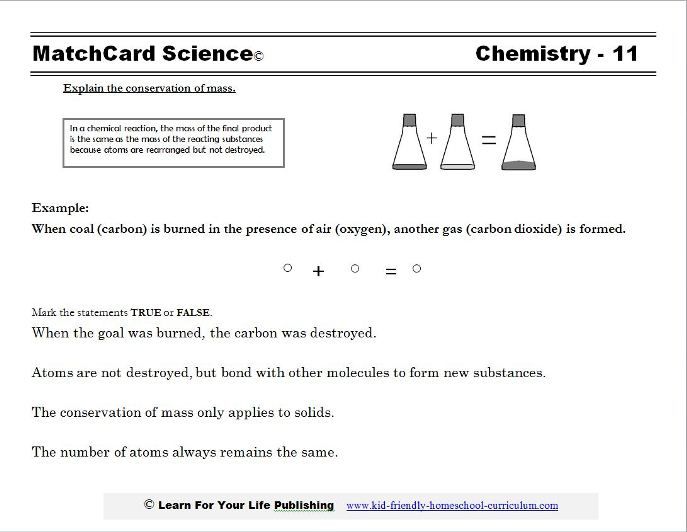
Defitinition: The Law of Conservation of mass is a science principle that says the in any closed system subjected to no external forces, the mass is constant irrespective of its To start the lab, we first recieved an alka seltzer tablet and a cup a water. We measured the tablet
Definition of Conservation of Mass: The mass of the products in a chemical reaction must equal the mass of the reactants. Explanation: Consider the reaction where hydrogen and …
The law of conservation of mass states that matter is neither created nor destroyed during a chemical reaction. Therefore, the mass of a system should remain constant during any chemical process. In this experiment, you will determine whether mass is conserved by examining a simple chemical reaction
1/10/2012 · This is a chemistry video for Grade 10-11 students that demonstrates the Law of Conservation of Mass or Law of Conservation of Matter with an experiment. This is a chemistry video for Grade 10-11
Conservation of Mass Lab 3. If the water bottle did not have a tight cap, would it be possible to prove the law of conservation of mass in this lab? 4. When you removed the cap from the bottle, what was the noise you heard? 5. Explain why there may have been a difference in mass after you removed the cap from the bottle. Title: Conservation of Mass Author: KSD Created Date: 4/16/2014 2:00
29/11/2011 · A closer look at the Law of Conservation of Mass through the use of an experiment, and a model! Find more at www.jbsedweb.weebly.com ! Find more at www.jbsedweb.weebly.com ! Category
In the lab, we are going to see if the mass of the compounds we start with match the mass of our reactants after the reaction takes place. Safety: Wear safety goggles and a lab apron for this activity, you are handling a weak acid!
Experiment #8 – Types of Reactions and Conservation of Mass Objectives: To become familiar with several different types of reactions. To study the law of
Either the Law of Conservation of Mass was not true in beta decay, or there was another unknown, and, at that time, undetectable particle, which was released and ultimately responsible for the “loss” of mass. In the process of doing this lab, students will see the value of accurate measurements and the use of the scientific method when confronted with an apparent contradiction in basic
This assessment has two multiple choice questions and two short response questions about the Law of Conservation of Mass/Matter. Students need to know what the law states and have a good understanding about what happens to atoms during a chemical change.
Summary. In this lab, students perform three simple experiments, recording the mass of a substance before and after a reaction. They then explain how the law of conservation of mass was not violated, even if the mass changed.
Classroom Resources Conservation of Mass AACT

Conservation of mass Wikipedia
Law of conservation of mass: matter cannot be created or destroyed in a chemical reaction or physical transformation. Implies that during any reaction in a closes system, the mass of the reactants must equal the mass of the products.
2) Using ½ Alkaseltzer tablet, a zipper baggie & any other lab equipment you need (ask your teacher), please prove the Law of Conservation of Mass. Call your teacher to your lab table when you have proved it before you dispose of your lab materials.
Name:_____ Conservation of Mass Lab SNC2D Question: Does the mass of the reactants differ from the mass of the products in a chemical
Lab 12: Law of Conservation of Mass Inquiry Name: _____ Date: _____ Period: _____ Purpose: To design and complete a laboratory experiment that proves the law of conservation of mass.
Law of Conservation of Mass – Experiment 3. The experiment will explore whether matter is created or destroyed during a chemical reaction.
Therefore this lab was to verify the law of conservation of mass using vinegar and baking soda. In this lab, students work in small groups of two to complete a laboratory exercise related to conservation of mass. Using vinegar, baking soda, and a 250ml beaker, students measured the amount of gas that was released or in escaped in an open system (not sealed). Students then seal the vinegar and
According to the Law of Conservation of Mass, the mass of metallic copper reclaimed at the end of the series should be the same as the initial mass. 296 Lab Manual Conservation of Mass
Conservation of Energy Physics Lab VI Objective This lab experiment explores the principle of energy conservation. You will analyze the flnal speed of an air track glider pulled along an air track by a mass hanger and string via

substance by a mass equal to the amount of oxygen that reacted with the substance. These experiments led to what is currently known as The Law of Conservation of Mass. This law states that mass can neither be created nor destroyed.
conservation law comes out of Newtonʹs second law. Remember that the gravitational force on the Remember that the gravitational force on the falling ball is the constant mg, where m is the mass of the ball and g is a constant acceleration due to
The law of conservation of matter states that in any given system that is closed to the transfer of matter, the amount of matter in the system stays constant Answer b The law of conservation of matter says that in chemical reactions, the total mass of the products must equal the total mass …
Either the Law of Conservation of Mass was not true in beta decay, or there was another unknown, and, at that time, undetectable particle, which was released and ultimately responsible for the “loss” of mass.
Thus, the Law of Conservation of Mass is verified. Precautions: When setting up the apparatus, and while taking the first reading of the total mass of apparatus and reactants, care must be taken that the barium chloride solution in the ignition tube does not spill and mix with the sodium sulphate solution in the conical flask.
Conservation of Mass Lab Data Questions 1. What is the precision of the triple beam balance? The precision of the triple beam balance is 0.1 (1/10th) grams. (That is the smallest division of measurement on it. ***In order to conclude that there was a mass change, the difference must be greater than the precision of the triple beam balance and no errors to explain the change*** Data …
The law of conservation of mass or principle of mass conservation states that for any system closed to all transfers of matter and energy, the mass of the system must remain constant over time, as system’s mass cannot change, so quantity cannot be added nor removed. Hence, the quantity of mass is conserved over time.
2 Abstract The purpose of this lab is to verify the law of conservation of energy. The law of conservation of energy states that in an isolated system energy cannot be
Lab Section M Tu W Th F Chemistry 130 Experiment: Conservation of Mass Our experiment this week will again investigate mass changes in relationship with a chemical reaction. The Law of Conservation of Mass states that “matter is neither created nor destroyed in a chemical reaction.” We are following this idea when we “balance” our equations, by being sure that the number and kinds of
Law of Conservation of Mass Lab – Download as Word Doc (.doc), PDF File (.pdf), Text File (.txt) or read online. Scribd is the world’s largest social reading and publishing site. Search Search
Conservation of Mass Lab The Law of Conservation of Mass states that mass in a closed system will remain constant, regardless of the processes acting on the system. A closed system is one to which nothing can be added or taken away. Procedure: 1. Record the mass of an empty beaker using the digital scale or triple beam balance. 2. Add a set volume of water to the beaker (record the volume of
Law of Conservation of Mass Lab Purpose: To attempt to verify & observe the Law of Conservation of Mass – In any chemical reaction, the total mass of the reactants is always equal to the mass …
Maintaining Mass page 3 3. The student will be able to correctly hypothesize and modify the provided demonstration in a way that will make it an accurate presentation of conservation of mass.
1. Always check to confirm that the balanced is tared before making mass measurements. 2. Add 50. mL of water to a clean Erlenmeyer flask. 3.
Name:_____ Period:_____ Lab 2-Investigating the Law of Conservation of Mass Objective: To corroborate the law of conservation of mass through laboratory
Name Period Conservation of Mass Lab
12/08/2018 · The Law of Conservation of Mass indicates that mass cannot be created nor destroyed. This means that the total mass of the reactants in a chemical reaction will equal the total mass …
Stoichiometry: Baking Soda and Vinegar Reactions Teacher Version In this lab, students will examine the chemical reaction between baking soda and vinegar, and mix different amounts of these household chemicals to learn about the concept of stoichiometry. California Science Content Standards: • 3. Conservation of Matter and Stoichiometry: The conservation of atoms in chemical reactions …
Collisions and conservation laws Goal: The momentum of an object is p = mv, where p represents the momentum, m represents the mass, and v represents the velocity. Note that momentum and velocity are vector quantities but since the entire lab is in one dimension the vector symbols will be dropped throughout the lab. Conservation of momentum. The conservation of momentum states …
Laboratory Experiment: Conservation of Mass Chemistry Standard 3 Introduction “Plop, plop, fizz, fizz, oil, what a relief it is:’ claims an old television ad for a popular antacid.
Consider a body of mass m, being accelerated by a compressed spring. As you veri ed in the rst As you veri ed in the rst experiment of the semester, the force exerted by a spring is given by Hooke’s Law, F = …
Conservation of Mass Lab In this laboratory activity, the conservation of mass will be proven. There will be two trials using the same amount of each material. The first trial is an open system. The students will decide if an open system confirms the Conservation of Mass, which states that matter can neither be created nor destroyed only transformed. The second trial will be using a closed – law and justice in australia prue vines pdf Name _____ Period _____ Points _____/15 Lab: Law of Conservation of Mass Objective: The purpose of this law is to prove the law of conservation of mass using a very simple
Conservation of Mass and how it is applicable to everyday life /10Outlines the purpose of the lab and explaining its importance Includes a hypothesis using the proper methodology for a hypothesis
In this lesson students will examine the Law of Conservation of Mass in a chemical reaction, showing the total mass of the reactants equals the mass of the …
of conservation of mass. In this lab, the goal is to try and prove this law by In this lab, the goal is to try and prove this law by experimenting with vinegar and baking soda, a simple chemical reaction.
The Law of Conservation of Mass Lab Atomic Concepts Hannah Pre-Lab Discussion Matter cannot be created nor destroyed in a chemical change. This principal, first proposed by Antoine Lavoisier in the mid 1700’s, is known as the Law of Conservation of Mass. This law applies to all chemical reactions. During a chemical change, atoms rearrange themselves but are not created or destroyed. In this
an experiment with a different procedure to show the law of conservation of mass using an Alka-seltzer tablet and water (write this out on a separate sheet of paper).
(d)Mass can neither be created nor destroyed as per the law of conservation of mass. (c)(23 x 2) +16 = 62. (b)The balanced chemical equation satisfies the laws of conservation of mass.
Name Period Points /15 Lab Law of Conservation of Mass
Lab Alka-Seltzer Rockets American Chemical Society

Law of conservation of mass (experiment) YouTube
Lab 7 Collisions and conservation laws
Law of Conservation of Mass Lab Report.doc Scribd
Law of Conservation of Mass experiment Law of


The Law of Conservation of Mass Newfane Central School
Conservation of Mass Worksheet Indianapolis Public Schools
law and order season 15 episode guide – Lab 2-Investigating the Law of Conservation of Mass
Law of Conservation Of Mass-Lab Report by isaiah irizarry

NCERT Class 9 Science Lab Manual – Law of Conservation of Mass
Chemistry 130 Experiment Conservation of Mass
Raptor Science LAW OF CONSERVATION OF MASS (LAB)
NCERT Class 9 Science Lab Manual – Law of Conservation of Mass
Name:_____ Conservation of Mass Lab SNC2D Question: Does the mass of the reactants differ from the mass of the products in a chemical
Conservation of Mass Lab The Law of Conservation of Mass states that mass in a closed system will remain constant, regardless of the processes acting on the system. A closed system is one to which nothing can be added or taken away. Procedure: 1. Record the mass of an empty beaker using the digital scale or triple beam balance. 2. Add a set volume of water to the beaker (record the volume of
According to the Law of Conservation of Mass, the mass of metallic copper reclaimed at the end of the series should be the same as the initial mass. 296 Lab Manual Conservation of Mass
Summary. In this lab, students perform three simple experiments, recording the mass of a substance before and after a reaction. They then explain how the law of conservation of mass was not violated, even if the mass changed.
This assessment has two multiple choice questions and two short response questions about the Law of Conservation of Mass/Matter. Students need to know what the law states and have a good understanding about what happens to atoms during a chemical change.
The Law of Conservation of Mass Lab Atomic Concepts Hannah Pre-Lab Discussion Matter cannot be created nor destroyed in a chemical change. This principal, first proposed by Antoine Lavoisier in the mid 1700’s, is known as the Law of Conservation of Mass. This law applies to all chemical reactions. During a chemical change, atoms rearrange themselves but are not created or destroyed. In this
Law of Conservation of Mass Lab – Download as Word Doc (.doc), PDF File (.pdf), Text File (.txt) or read online. Scribd is the world’s largest social reading and publishing site. Search Search
Conservation of Energy Physics Lab VI Objective This lab experiment explores the principle of energy conservation. You will analyze the flnal speed of an air track glider pulled along an air track by a mass hanger and string via