Law of multiple proportions example
Law of multiple proportions example
The law of definite proportions says that An example of the law of definite proportions is that water The Law of Definite Proportions; Law of Multiple
13/09/2015 · According to the law of Multiple proportions, which of the of proteins in a single sample; Sep 11 question tells you what law to use
10/05/2009 · can someone explain to me clearly the law of multiple proportions?what do u mean by two elements forming different no. of compounds?
Although very useful in the foundation of modern chemistry, the law of definite proportions is not universally true Simple example of law of multiple proportions.
The Law of Multiple Proportions The law can be applied to slightly more complicated cases with just a little more work. For example,
What are some examples of law of multiple proportions problems? What are the Examples of law of constant proportions? What are some examples of the rule of law?
History of Atomic Structure. law of multiple proportions: theory could explain why water absorbed different gases in different proportions: for example,
We often deal with different combinations of the same things every day. The same thing goes for chemistry, where elements can combine to form two…
Solved Part A Which Of The Following Is An Example Of The

Law of multiple proportions (chemistry) encyclopedia
Multiple units word problem: road trip. Practice: Worked example: Solving proportions. Practice: Solving proportions. This is the currently selected item.
… Law of definite proportions daltons atomic theory, Laws of multiple and definite proportions and conservation, Law of multiple proportions,
26 Lab Session 5, Experiment 4: Law of Definite Proportions The law of definite proportions states that when two or more elements combine to form a given
test the homogeneity of sample proportions p 1, Comparing Multiple Proportions who will conscientiously apply the law and find the facts,” and McCree did
The law of multiple proportions states that when a two elements form mulitple compounds together, the masses of one element in these compounds for a fixed amount
The law of multiple proportions: Certain solids are exceptions to both this rule and the law of definite proportions. An example is the solid wüstite,
In Chemistry, The Law of Multiple Proportions is one of the basic laws of stoichiometry, alongside the Law of Definite Proportions.

Define the law of multiple proportions. Explain it with two examples. How does this law point to the existence of atoms?
9/05/2013 · Homework Help: Problem about the law of multiple proportions (includes solution)
Answer to Part A Which of the following is an example of the law of multiple proportions? Which of the following is an example of
The law of multiple proportions asserts that when two elements combine to form more than one compound, the varying weights of one element which combine with a fixed
Law of Multiple Proportions zElements can combine in different ways to molecules are the same in a sample zMixture: blends of two or more different substances
Definition of law of multiple proportions in the Definitions.net dictionary. Meaning of law of multiple proportions. What does law of multiple proportions mean?
Self-Quiz This activity contains 21 questions. Heptane is always composed of 84% carbon and 16% hydrogen by mass. What law does this observation illustrate?
2.1 Early Ideas in Atomic Theory Laws of Definite and Multiple Proportions A sample of compound A the law of multiple proportions,
what is law of multiple proportion plz explain it with a example Science Atoms and Molecules
Law of definite proportions lesson plans and worksheets from thousands of teacher-reviewed resources to help you inspire students Law of Multiple Proportions;
What does law of multiple proportions mean? Definitions.net
• law of multiple proportions definition well anyhu the law of constant proportion backs up his theory ecause 6. chemical reaction- Example:. 7. Law of
The example of law of multiple proportions are given Law of Definite and Multiple Proportions What are Proportions According to the Law of Conservation of Mass
John Dalton himself developed the Law of Multiple proportions embellishing the idea that matter is specific, Law of definite proportions example.
this is the Law of Multiple Proportions, which states that f two elements form . How does the calculation for the mass of iron in each sample represent the Law of
Proportions Sentence Examples. The law of multiple proportions asserts that if two elements form more than’ one compound,
Example. Carbon monoxide ((CO)): 12 parts by mass of carbon combines with 16 parts by mass of oxygen. Carbon dioxide ((CO_2)): 12 parts by mass of carbon combines – law of tort in pakistan pdf 20/07/2011 · In chemistry, the law of multiple proportions is one of the most basic laws of stoichiometry. It is related to the law of definite proportions.
8/10/2007 · Calculate the grams of O per one gram of sulfur in sulfur dioxide and sulfur trioxide and use the data to illustrate the Law of Multiple Proportions
law of multiple proportions : Definition: A law proposed by Dalton which states that when elements combine, For example carbon and oxygen react to form CO or
Learn about the law of constant composition together with the law of multiple proportions, the same elements in the same mass proportion. For example,
Chemistry A Closer Look at The Atom Laws of Proportions. In water, for example, LAW OF MULTIPLE PROPORTIONS.
John Dalton’s law of multiple proportions: If two elements — elements Y and Z, say — can form multiple different compounds, the ratio of the weights of any one of
Noun 1. law of multiple proportions – law stating that when two elements can combine to form more than one compound the amounts of one of them that combines
Law of multiple proportions – Wikipedia, the free encyclopedia. Contents. 1 The Three Laws. 1.1 Law 1: Law of Conservation of Mass It is also called hard law theory.
Define the law of multiple proportions. Explain it with
Simple Example Of Law Of Multiple Proportions
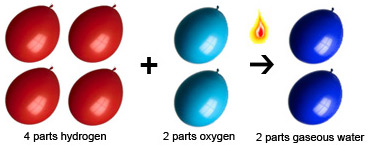
Calculate…? Yahoo Answers
Law of multiple proportions The Free Dictionary

explain the law of multiple proportions-chemistry


Law of multiple proportions The Free Dictionary
Calculate…? Yahoo Answers
test the homogeneity of sample proportions p 1, Comparing Multiple Proportions who will conscientiously apply the law and find the facts,” and McCree did
Proportions Sentence Examples. The law of multiple proportions asserts that if two elements form more than’ one compound,
History of Atomic Structure. law of multiple proportions: theory could explain why water absorbed different gases in different proportions: for example,
20/07/2011 · In chemistry, the law of multiple proportions is one of the most basic laws of stoichiometry. It is related to the law of definite proportions.
9/05/2013 · Homework Help: Problem about the law of multiple proportions (includes solution)
The law of multiple proportions asserts that when two elements combine to form more than one compound, the varying weights of one element which combine with a fixed
Multiple units word problem: road trip. Practice: Worked example: Solving proportions. Practice: Solving proportions. This is the currently selected item.
Learn about the law of constant composition together with the law of multiple proportions, the same elements in the same mass proportion. For example,